The Advanced Light Microscopy core at Penn State College of Medicine offers consultation and training for fluorescence imaging of molecules, cells, tissue and organs.
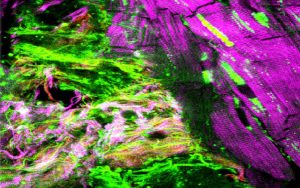 The Confocal, Super-Resolution STED and Deconvolution microscopes are useful in 3D imaging of any biological systems and spatial distribution of multiple fluorescently-labeled sub-cellular objects, including various cellular proteins complexes (multiple colors) simultaneously in live or fixed biological conditions.
The Confocal, Super-Resolution STED and Deconvolution microscopes are useful in 3D imaging of any biological systems and spatial distribution of multiple fluorescently-labeled sub-cellular objects, including various cellular proteins complexes (multiple colors) simultaneously in live or fixed biological conditions.
For fast volumetric imaging of cleared tissues and organs, the core also has a Miltenyi Biotec UltraMicroscope II.
High-end imaging workstations with licenses for Imaris and Huygens are excellent tools for image processing, analysis, and visualization.
Enter the Wild Onions Biological Imaging in the Life Sciences Competition!
Jump to topic
Search
Instrumentation and Services
Leica SP8 FALCON
The Leica SP8 FALCON (Room C1733) is an inverted laser scanning confocal microscope with fluorescence lifetime imaging (FLIM) and fluorescence correlation spectroscopy (FCS) capabilities.
- Software module: LIGHTNING for image enhancement
- Software module: Navigator for overview scans and subsequent acquisitions
- Tunable emission filter technology (AOBS) and tunable pulsed white light laser source
- Fully automated operational procedures
- High numerical aperture immersion objectives
- Differential interference contrast (DIC) imaging
- Line and area scanning capabilities with simultaneous or sequential scanning options
- Ultra-high-speed live cell imaging, complex cell tracking experiments, FRAP measurements, ratio metric experiments, complex FRET measurements,
- Tile scanning (scanning whole slide)
- Hybrid signal detection systems which allow 3D or 4D image generation at much lower light energies
- Time-gated fluorescence signal collection and single photon counting
- Environmental control via a Tokai Hit stage-top incubator facilitates live-cell imaging at physiological conditions (5% CO2 and 37C)
Leica SP8 STED 3X
The Leica SP8 STED 3X (Room C1730) is an inverted confocal/STED microscope which achieves super-resolution (down to 10s of nanometers) through the process of stimulated emission depletion.
- Three depletion lasers (592nm, 660nm, 775nm) matched with a pulsed white light laser for excitation allow for more flexible STED experiments
- 2D/3D STED
- STED is possible with either a 100x/1.4 oil objective or a 93x/1.3 glycerol objective
- For standard confocal imaging, a 20x/0.7 air objective, 63x/1.4 oil objective, and 63x/1.2 water immersion objective are also available
- FRET/FRAP software modules
UltraMicroscope II
The UltraMicroscope II from Miltenyi Biotec (C1733B) allows for fast 3D imaging of tissues and organs and is compatible with many clearing protocols. Four laser lines at 488nm, 561nm, 640nm, and 785nm allow for multiple combinations of fluorophores. Users can tweak the shape and number of light sheets in order to match their experimental goals.
DeltaVision Elite
 The DeltaVision Elite (Room C1728) is a reliable inverted widefield microscope with an acrylic enclosure for maximum stability during live-cell imaging experiments. Tailored deconvolution enhances image quality while maintaining data integrity for downstream quantitative analyses.
The DeltaVision Elite (Room C1728) is a reliable inverted widefield microscope with an acrylic enclosure for maximum stability during live-cell imaging experiments. Tailored deconvolution enhances image quality while maintaining data integrity for downstream quantitative analyses.
- Precise optical sectioning capability and deconvolution software
- Multi-wavelength switching excitation light source
- High numerical aperture immersion objectives
- Highly sensitive cooled CCD and EM-CCD cameras
- Differential interference contrast (DIC) imaging
- Tile scanning (scanning whole slide)
Commercially licensed software
We offer commercial software (Huygens and Imaris) for deconvolution and image visualization/analysis.

- Surface renderings
- Deconvolution
- Segmentation
- Fluorescence intensity measurements
- Complex cell tracking measurements
- Voxel counting
- Batch processing
- Drug bio-distribution measurements
- Accurate colocalization analysis
- Many other complex image processing, statistical calculations depending upon researcher’s needs
Imaris Workstation
The Imaris Workstation (Room C1732A) is equipped with a 24-core CPU, 512GB of RAM, an NVIDIA 4090 GPU, and 8TB of NVMe storage. It is capable of handling large datasets with licenses for Imaris and Huygens Professional. Other software include Fiji, ilastik, Qupath and Napari.
This image analysis workstation is available for independent use with a low hourly rate, but training/assisted sessions are also available.
Reach out if you’re interested in using deep learning tools such as stardist, cellpose, Noise2Void, etc.
Specifications
- CPU: 24 core AMD Ryzen Threadripper PRO 5965WX, 3.80 GHz with 128MB L3 Cache
- RAM: 512 GB (8 x 64 GB), ECC DDR4
- GPU: NVIDIA GeForce RTX 4090 with 24GB of VRAM
- OS drive: WD Black 4TB SN850X NVMe drive
- Data drive: WD Black 4TB SN850X NVMe drive
Procedures, Protocols and Forms
The instruments in Advanced Light Microscopy are very sensitive and enormously expensive. These instruments must be treated with care by all users, as improper or careless use could render these systems unusable for extended periods of time, impacting multiple research programs. Even under service contract, many things will take weeks to get fixed, and service contracts do not cover negligence on the part of the users. Thus, to keep all of the equipment functioning properly for everyone, the following rules apply to all jobs done on the shared microscopes. These rules and regulations are subject to change at any time.
- No foods are allowed in the microscope rooms at any time.
- No sitting/standing on the anti-vibration table.
- No installing software onto the computer.
- No using the computers for purposes unrelated to microscopy (for instance, checking email, surfing internet, etc.)
- The microscope room is equipped with Class IIIb and Class IV lasers. Therefore, unauthorized entry into the facility is prohibited at all times.
- Before training, please arrange a 15-minute appointment to determine the applicability of the facility equipment to your research goals.
- Fill out a project survey form.
- Users need to attend three hours of training before being allowed to sign up to use the equipment independently. Additional collaborative opportunities, including image processing, image analysis and data interpretation, etc., are available for those who need it.
- Refresher training may be required for anyone who has not used the facility equipment for six months.
- Users may sign up for use of the microscopy equipment between 8 a.m. and 6 p.m. weekdays. After-hours and weekend appointments may be made by arrangement with the Facility Manager.
- You may schedule equipment time up to six days in advance. See scheduling links.
- Cancellation must be given at least 24 hours in advance of the requested time slot, preferably sooner. Failure to cancel in advance will result in the following penalties:
- First offense: a warning
- Second offense: time slot missed billed in full
Investigators who must cancel who can find someone to fill in the canceled time will receive no penalty.
- Investigators finishing early must contact the next person signed up.
- Signup slots may be cancelled by the facility staff at any time to schedule needed maintenance or repairs.
- Users must include their phone number and email during online signup.
- In the login book, users must also include lasers, PMTs, etc., that will be used.
- Use of the facility is a privilege. Access to confocal computer system will be activated only after successful completion of all training sessions. Investigators will use their Penn State Health ePass ID and password for login, once those have been activated following successful completion of training.
- Users must follow the standard operating procedures (SOP) when operating the instruments. If unsure of any procedures, users should contact the Facility Manager for further information.
- Any misuse of the facility will result in immediate prohibition.
- Investigators should check the schedule to see if someone else is signed up after them. Follow the Shutdown Procedure included in the standard operating procedures. Make sure all equipment is cooled down and turned off properly. Each investigator is responsible for logging off properly to avoid extra time charges, and for turning off all equipment.
- Users are responsible for cleaning up the microscope tables upon completion of imaging work.
- Slides, coverslips, specimens, and other tools must be removed from the microscopes, and the air table should be cleaned up. Users must wipe oil/water with lens tissue or Q-tips from all oil/water objectives used.
All publications, press releases or other documents that result from the utilization of any Penn State College of Medicine Institutional Research Resources including funding, tools, services or support are required to credit the core facility and associated RRID for each core used. Use of an instruments in the Advanced Light Microscopy core should include the following:
“The Advanced Light Microscopy core (RRID:SCR_022526) services and instruments used in this project were funded, in part, by the Pennsylvania State University College of Medicine via the Office of the Vice Dean of Research and Graduate Students and the Pennsylvania Department of Health using Tobacco Settlement Funds (CURE). The content is solely the responsibility of the authors and does not necessarily represent the official views of the University or College of Medicine. The Pennsylvania Department of Health specifically disclaims responsibility for any analyses, interpretations or conclusions.”
Safety for all users and Core personnel is a top priority. Since the facility is a shared laboratory and is designated as a Biosafety Level II facility, there are certain guidelines that all users are responsible to be aware of and follow. This policy includes guidelines for the safe use of lasers as well as procedures for imaging any biohazards and proper cleanup procedures. Any imaging of cells or materials classified with Biosafety Levels 2 or 2+ (including but not limited to live primary cells, cells containing pathogenic viruses) must be approved by the Institutional Biological Safety and Recombinant DNA Committee, an SOP and the BS number supplied when booking time.
Not all BL2+ work will be approved; no Biosafety classification work above BL2+ is ever accepted.
Rules for the Safe Use of the Confocal Microscopy Facility
The Confocal Microscopy Facility operates as a Biosafety Level 2 laboratory. These rules are reviewed and revised as necessary each year, and each facility user is responsible to review them on a regular basis to insure they are in compliance. Briefly, the rules for safe use of BSL-2 materials as applied specifically to the Confocal Microscopy Facility are as follows:
- Admission to the laboratory is restricted to authorized personnel when work with human samples or known infectious agents is in progress.
- All human, primate, biohazardous or genetically modified cells must be assigned appropriate BSL levels for research use by the Biological Safety and Recombinant DNA Committee before they are allowed to be used in the Confocal Microscopy Facility. See the Biohazardous Materials, Use in Research and Instruction policy for details (internal access only; ePass login required).
- All samples that are intended for imaging and all unfixed biohazardous cells intended for analysis must be clearly identified and approved in advance by the Microscopy Imaging Safety Committee. Generally this is covered by the above process of evaluation by the Biological Safety and Recombinant DNA Committee.
- In general, unfixed primary human and primate cells will not be allowed to be imaged on the Confocal, although some exceptions may be made if the cells have been certified as biohazard/pathogen-free. Any need to image unfixed cells must be justified and pre-approved in writing by the Core Facility Manager.
- All procedures must be performed carefully to minimize the creation of aerosols.
- All samples must be logged onto the facility in the logbooks provided. Samples that have been fixed for imaging on the Confocal microscopes may be monitored and recorded to ensure that the fixation is adequate.
- Work surfaces are to be decontaminated with 10 percent household bleach after any spillage of sample or hazardous material and after each user completes their work.
- Instrument decontamination shall be performed according to protocols posted on each instrument, an addition to any special procedures required for particular samples (as decided by the Microscopy Imaging Safety Committee).
- All spills, however small, must be recorded in the accident book and reported to the Core Facility Manager.
- All liquid and solid waste and disposal is to be removed from the laboratory as biohazardous material by each user.
- Gloves must be worn whenever handling or running biohazardous samples on the microscope.
- Hands must be washed after handling biohazardous materials and before leaving the laboratory.
- Regardless of biosafety level, thorough cleanup of the microscope – including oil-immersion and other lenses – and the sample stage and other surfaces, is required after every imaging session. Failure to clean up properly will first result in a warning; continued failure to leave the microscope in a completely useable, ready state for the next researcher will result in loss of privileges on the microscope.
- Neither syringes nor hypodermic needles should be used in the facility without prior authorization. This consent must be obtained each time syringes or hypodermic needles are used; consent for use today does not imply that consent is given for later dates.
- No eating, drinking, smoking or applying cosmetics in the work area.
- No mouth pipetting.
- No animals are permitted within the laboratory.
In addition to the above guidelines and because this facility uses Class III, the following Laser Safety guidelines shall also be observed:
- Access to the laboratory will be limited to only those persons necessary to the running of experiments when any laser is operating.
- Only qualified personnel shall operate or adjust any laser.
In the event of a potential retrovirus exposure
This item refers to exposure of skin or mucous membranes to infectious materials and is modeled after the NIH program “3 Emergency Steps to Take in the Event of a Potential HIV Exposure.”
Remember that the risk of infection is very dependent upon the titer of virus and the route of exposure. Thus the risk of infection by contact of intact skin with infectious body fluids is probably truly zero, and although the risk of infection by contact of mucous membranes or non-intact skin with infectious body fluids may be extremely small, it is likely to be non-zero.
The emergency steps are:
- Immediately initiate first aid at the work site.
- Contaminated skin should be meticulously cleaned for 10 minutes using a povidone iodine solution (such as Betadine) and copious amounts of water.
- Contaminated eyes and mucous membranes should be irrigated for five minutes using water.
- Notify your supervisor, if they are immediately available.
- Report to the hospital emergency department to activate the invasive accident protocol (needlestick protocol), which will include evaluation, counseling and provision of antiretroviral treatment if deemed appropriate. Do this immediately (within the half-hour).
- In the event of an emergency (e.g., fire, water leaks), investigators should act calmly and ensure their own personal safety first. If possible, shut down all running equipment, cover equipment if possible, and exit the building.
- Notify the Core Director immediately and phone the campus emergency line at 717-531-8888 (on-campus, dial 8888).
Work with the Advanced Light Microscopy Core
For details on fees, sign in to iLab.
To reserve time on one of the light microscopes, sign in to iLab.
To use iLab, you must have an account, be assigned to a PI’s lab and have funding identified before you will be able to reserve time and use the microscope. See core staff for questions.
All potential users of light microscopes are required to have completed training prior to using the microscopes. See training requirements and find out how to schedule training.
